 
In the present work we analyzed in some detail the possible implication of the rafts-like microdomains in the fusion process. In previous works we have shown the pivotal role of these microdomains in fission machinery since the function of some proteins involved in mitochondria fission, such as DLP1, could be restricted to their localization in these structures. These structural platforms are not confined to the plasma membrane, but are similarly formed at subcellular organelles, which include endoplasmic reticulum, Golgi and mitochondria, named raft-like microdomains, or more shortly, “lipid rafts”. The structure of lipid rafts is dynamic, resulting in an ever-changing content of both lipids and proteins. Lipid rafts are membrane microdomains enriched in cholesterol and sphingolipids, which play many important roles in cell signal transduction. In fact, 3-(2,4-Dichloro-5-methoxyphenyl)-2,3-dihydro-2-thioxo-4(1H)-quinazolinone (Mdivi-1), a selective inhibitor of mitochondrial fission that acts by inhibiting the GTPase activity of DLP1, actually induces mitochondrial fusion in a well-organized network. Since mitochondria fusion and fission are strictly related, the inhibition of one of these processes leads to an increase of the other: for instance, the inhibition of DLP1-mediated fission causes unopposed mitochondrial fusion. Disruption of mitochondrial dynamics is emerging as a pathogenetic determinant in prevalent diseases. The balance between fission and fusion is very important for mitochondrial participation in crucial cellular processes: fission is required for mitosis, apoptosis, and autophagy, while fusion is a necessary adaptation to nutrient starvation and increased metabolic demand allowing transmission of transmembrane potential along interconnected mitochondria. Interaction of DLP1 with hFIS1 and mitochondrial fission factor (MFF1), a further molecule involved in the regulation of DRP1 activity, forms an oligomeric ring dividing mitochondria at distinct sites. The opposite process, mitochondria fission, is mediated, among others, by the dynamin-like protein 1 (DLP1), also called dynamin-related protein-1 (DRP1), which is recruited to the mitochondrial outer membrane, and, in human cells, by the human homologue of mitochondrial fission protein (hFIS1), initially described in yeast. 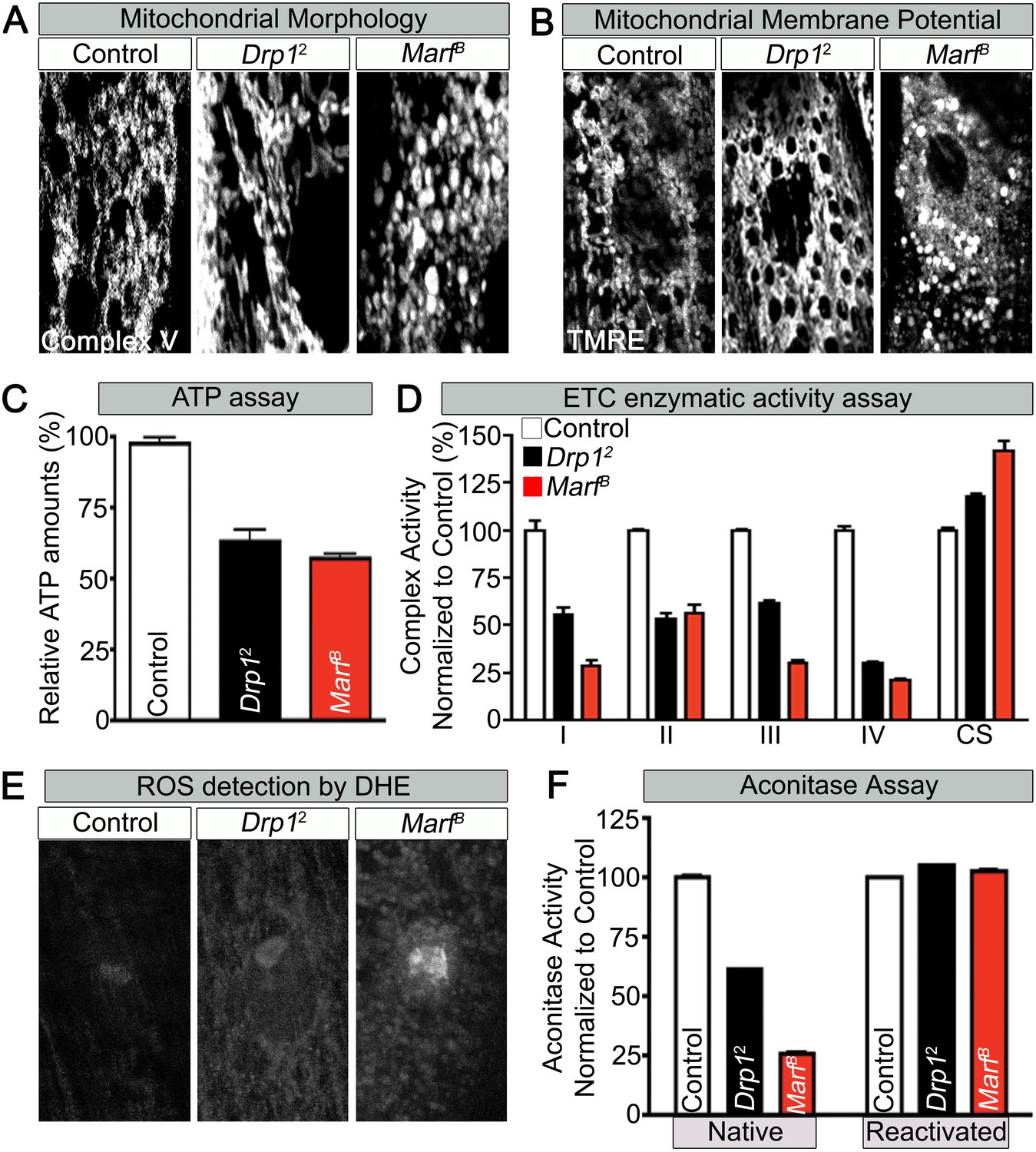
Mitochondrial fusion machinery is controlled by three guanosine triphosphate (GTPase) proteins: mitofusin 1 (MFN1) and mitofusin 2 (MFN2) that mediate fusion of outer membrane, and optic atrophy protein 1 (OPA1) that carried out fusion of inner membrane. This balance is of great relevance in both cell life and death since the mitochondrial network is involved in all the main cell activities such as metabolism, proliferation, autophagy and apoptosis. The mitochondrial dynamics describe the continuous changes in the position, size and shape of mitochondria within cells, consisting in a balance of fusion and fission pathways, with each process mediated by a distinct set of interacting factors. Mitochondria are a highly responsive network of organelles in constant balance between two states: a set of individual spherical organelle or an elaborate network of interconnected elements. Our results underscore the possible implication of lipid “rafts” in mitochondrial morphogenetic changes and their homeostasis. In the present work we investigated the implication of lipid rafts in mitochondrial fusion induced by Mdivi-1. 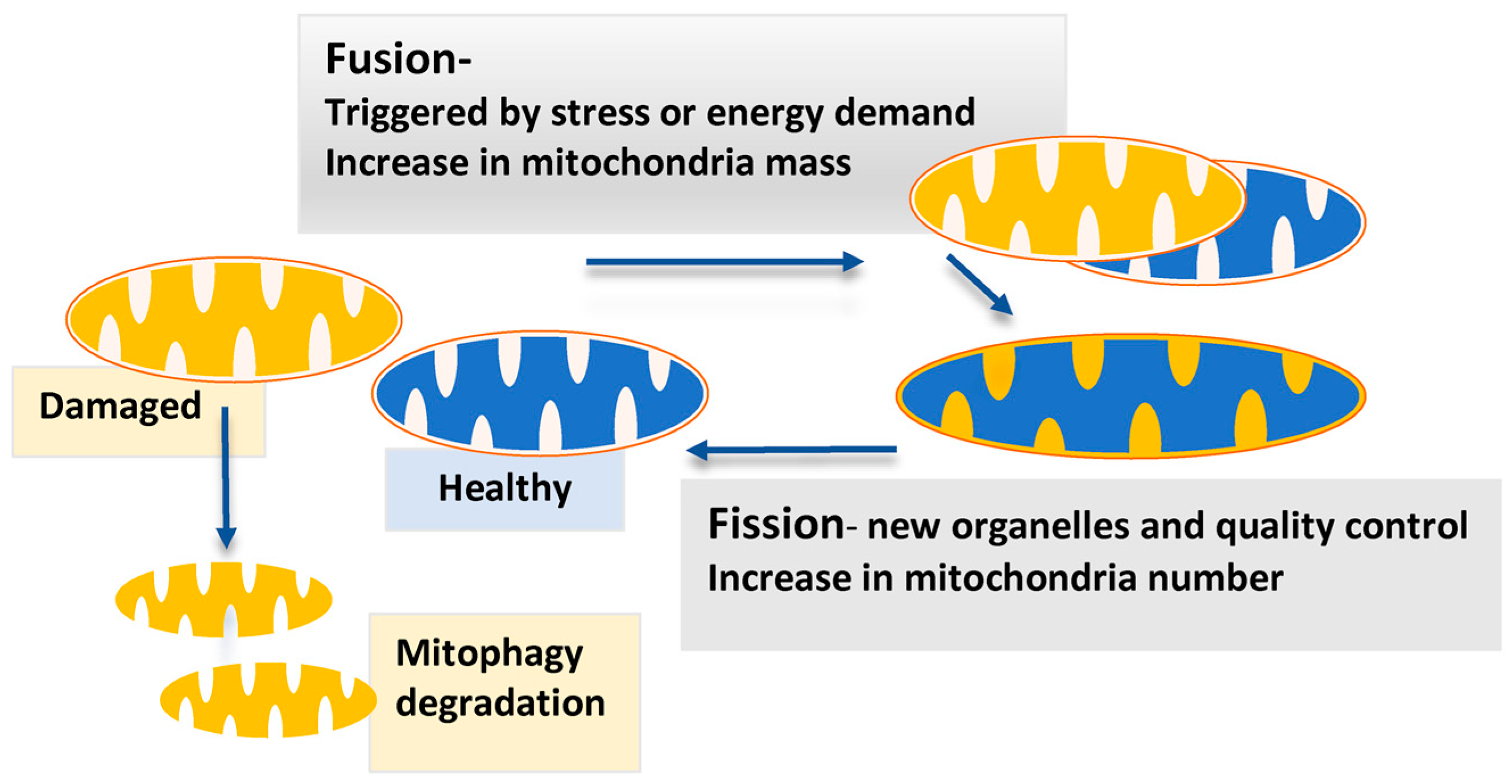
Mitochondrial fusion and fission machinery is controlled by key proteins such as mitofusins, OPA-1 and several further molecules. The regulation of the mitochondrial dynamics and the balance between fusion and fission processes are crucial for the health and fate of the cell. Received: JAccepted: FebruPublished: April 10, 2018 Keywords: lipid rafts mitochondria fusion apoptosis reactive oxygen species #These authors to be considered as Senior Authors *These authors contributed equally to this work Laura Ciarlo 1, *, Rosa Vona 1, *, Valeria Manganelli 2, *, Lucrezia Gambardella 1, Carla Raggi 3, Matteo Marconi 1, Walter Malorni 1, Maurizio Sorice 2, Tina Garofalo 2, # and Paola Matarrese 1, 4, #ġOncology Unit, Center for Gender-Specific Medicine, Istituto Superiore di Sanità, Rome, ItalyĢDepartment of Experimental Medicine, Sapienza University, Rome, ItalyģCenter for Behavioral Sciences and Mental Health, Istituto Superiore di Sanità, Rome, Italy 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
